Convenient Routes to Efficiently N-PEGylated Peptides
Christian Stutz 1, Anna Meszynska 2, Jean-François Lutz 2, Hans G. Börner 1
1 Department of Chemistry, Laboratory for Organic Synthesis of Functional Systems, Humboldt-Universität zu Berlin, Brook-Taylor-Str. 2, Berlin 12489, Germany, E-mail: ;
2 Precision Macromolecular Chemistry Group, Institut Charles Sadron, UPR-22 CNRS, 23 rue de Loess, 67034 Strasbourg, France, E-mail:
Two modern supports are evaluated for being high efficient routes to N-terminal PEGylated peptides and compared to an established PS resin (Figure 1). 70 nm superparamagnetic core-shell nanoparticles with magnetite cores and amino functional silica shell proved to constitute a useful colloidal support for sequential assembly of peptides. Alternatively, end-functional poly(styrene) a recent development of soluble supports have been investigated, where peptide assembly takes place homogeneously in solution. The supports enable ease of conjugation of PEG with Mn= 5000 as important for pharmacological applications. Where Mn= 2000 leads to quantitative conjugation reactions with only 1.15 equiv. excess of the polymer with respect to the supported peptides, Mn= 5000 shows efficient coupling, leading to about 40% conversion. The supports investigated do not suffer from inherent diffusion limitations as compared to commonly established poly(styrene) microgel resins and thus prove particularly to be of value for coupling of larger residues e.g. PEG with Mn= 5000.
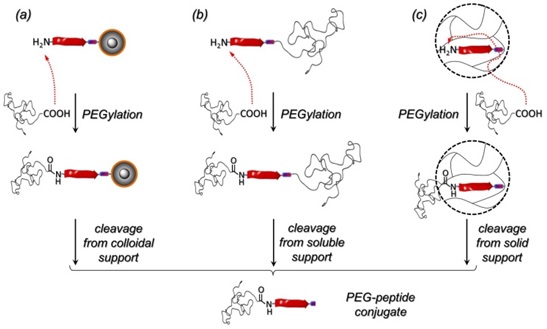 Figure 1. Strategies for N-terminal PEGylation of oligopeptides. Colloidal supports (a), soluble supports (b) and standard PS resins (c).
|