|
|

|
|
Prof. Dr. habil. Hans Börner
| Phone: |
+49 (0)30 2093-7348 |
| Fax: |
+49 (0)30 2093-7500 |
Email
|
|
|
|
|
|
|
|
Contact Us |
|
|
|
Laboratory for Organic Synthesis of Functional Systems
Department of Chemistry
Humboldt-Universität zu Berlin
Brook-Taylor-Str. 2
12489 Berlin
Germany
Sekretariat
Phone: +49 (0)30 2093-7349
Fax: +49 (0)30 2093-7215
Room: 0'144
Email
office.functional-systems hu-berlin.de |
|
|
|
|
|
|
News
Adding Spatial Control to Click Chemistry: Photo triggered DielsAlder Surface (Bio)functionalization at Ambient Temperature
A photoconjugation strategy based on light-triggered Diels–Alder addition of o-quinodimethanes is described. The method provides spatial control, is compatible with biomolecules, and proceeds rapidly at ambient temperature without the need of a catalyst.The photoenol-mediated conjugation strategy possesses many interesting features, making it a first-class click reaction. It can proceed rapidly at ambient temperature in a wide range of (polar) solvents. No catalyst is required and no by-product is formed, and thus no purification steps are required. Most importantly, control over time and space is inherent owing to its light-induced nature. Herein, we have demonstrated the high efficiency and speed of the phototriggered Diels–Alder (bio)conjugation in solution as well as on a surface by means of the covalent attachment of three dienophiles: a smallmolecule ATRP initiator, a polymer, and a peptide. We envisage many applications for this procedure ranging from the photolithographic grafting of conductive polymers for LED development to the generation of a variety of complex architectures, including polymer–protein conjugates. Our efforts are currently directed towards the production of functional patterned substrates to control cell behavior by the introduced technique.
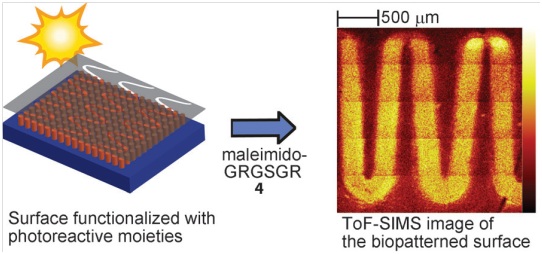
The paper results from a DFG funded collaboration with the research group of Christopher Barner-Kowollik from Karlsruhe Institute of Technology (KIT).
|
|
|